FAQ
Purchasing Math Illustrations
- Do you offer a bulk purchase discount?
- Do you offer a site/institutional license?
- Can I order by check/purchase order?
- What's the Difference between Math Illustrations and Geometry Expressions?
Yes! Here is the breakdown:
| Copies | Price |
|---|---|
| 1-4 | $59 |
| 5-9 | $55 |
| 10+ | $50 |
The discount will be automatically applied when you check out.
Yes! You can order a site license for an entire school/organization. The cost for a single OS (Windows or Macintosh) is $600. For both Windows and Macintosh the license is $750. This allows you to install Math Illustrations on as many computers at the site as you wish. Each teacher is also allowed to install Math illustrations on one home computer but can only use it for activities related to the purchasing school/organization.
We offer a multi-site discount so for multiple sites or to license Math Illustrations for an entire school district please contact us for a quote.
Currently we do not accept orders for site licenses online. Please fill out the following order form and fax or mail it to us at the following address:
P.O. Box 238755
Tigard, OR 97281
Voice: 503-968-6251
Fax: 503-968-1282
If this is not convenient please contact us and we can work something out!
Sure! You can fill out this order form and either fax or mail it to us. Here is our address:
P.O. Box 238755
Tigard, OR 97281
Voice: 503-968-6251
Fax: 503-968-1282
Geometry Expressions adds the power of symbolic calculations and many additional export formats to Math Illustrations' geometry-drawing base.
View the complete feature comparison chart here
Using Math Illustrations
- What are the system requirements to run Math Illustrations?
- When I start Math Illustrations it says "Cannot connect to server." What does that mean?
- How do I get my drawings into Word/PowerPoint?
- How do I distinguish between constraints and annotations?
- What does "over constrained" mean and how do I avoid it?
- Are there any precreated figures for Math Illustrations?
- I can't copy and paste from Math Illustrations to Microsoft Office 2004 on my Macintosh
- Run Preview (found in the Applications folder)
- Do a copy in Math Illustrations
- In Preview select "New From Clipboard" in the "File" menu
- In Preview select "Copy" from the "Edit" menu
- Paste into Office 2004
Windows
Windows 10 or later
Macintosh
OS X 11.0 or better.
The first time you run Math Illustrations it must authorize your key by contacting our server. If you receive this message it means Math Illustrations was not able to reach our server. Please check that your internet connection is working and any firewall software that would block an application from talking to the internet is turned off (or at least make an exception for Math Illustrations). It is also possible that our server is down (if you can reach this website then our server is up). If you are still having problems write us an email and we will sort it out.
Getting drawings into Word and PowerPoint is easy. Once you have your drawing the way you like it, choose the "Copy Rectangle"  button from the toolbar. Click and drag to draw a rectangle around your figure.
When you let go of the mouse button the figure will be placed on the clipboard. Now go to Word/PowerPoint and choose Edit->Paste (or press ctrl+v) and your drawing will appear in your document.
button from the toolbar. Click and drag to draw a rectangle around your figure.
When you let go of the mouse button the figure will be placed on the clipboard. Now go to Word/PowerPoint and choose Edit->Paste (or press ctrl+v) and your drawing will appear in your document.
By default constraints and annotations are set up to be slightly different colors. However, when creating a document it may be preferable for both constraints and annotations to be the same color. If this is the case, you can click on the toolbar button  to show a distinguishing mark next to constraints, annotations and measurements:
to show a distinguishing mark next to constraints, annotations and measurements:
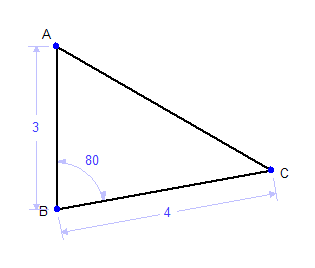
When dealing with constraint based systems you can sometimes get into a situation where you have too many constraints on a figure. For example, take the following triangle that has a constrained angle of 80° and two sides of length 3 and 4:
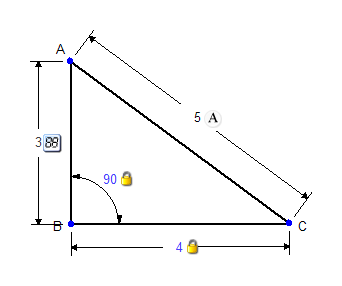
Now let's say that you want to constrain AC to length 5. Trying to do this will result in an overconstrained triangle. Setting AC to 5 would force the constrained angle to be 90°. In other words, the constraint engine cannot satisfy both the constraint on AC to be 5 and the constraint on the angle to be 80°. Without removing either the angle constraint or the distance constraint the triangle cannot be properly constructed.
One solution would be to take a measurement of side AC. Alternatively, if you wanted to show a dimension for the third side of the triangle you could use an Annotation instead of one of the constraints. Annotations are simply labels -- they have no effect on constructing a figure and resolving its constraints, although they look like constraints and are applied in the same way.
There are many example files that come with Math Illustrations. These examples can be found in the Examples directory.
Use the N-gon tool  in the Drawing toolbox to quickly and easily draw regular polygons with any number of sides.
in the Drawing toolbox to quickly and easily draw regular polygons with any number of sides.
For even more examples, visit the figure library to download files of the regular polygons as well as other useful precreated figures.
Math Illustrations v1.x running on OS X 10.4 or later can copy and paste to Microsoft Office 2004.
Math Illustrations v2.0 requires OS X 10.6 or later for copy and paste to Microsoft Office 2004 to work.
The following work-around will allow copy and paste on OS X 10.4 and 10.5:
Videos
Tips (email newsletter archive)
- July 2011 - Transformations
- May 2011 - Exterior and interior angles, constraints and constructions, pictures
- April 2011 - 3D cone and cylinder, interactive whiteboard
- March 2011 - Piecewise functions and exterior angles
- January 2011 - Inequalities, number lines and normal distributions
- December 2010 - Venn diagrams, mapping diagrams and hyperboloids
- October 2010 - Functions with parameters, transforming functions and generic functions
- September 2010 - Geometry annotations, trig graph and regular polygon
Documents